 Thus these valence electrons are now easily available to take part in a reaction and the reactivity of the element is said to be increased. The outermost electrons become farther away from the nucleus.Īs a result, the shielding effect increases, and the valence electrons are more loosely bound to the nucleus. As we move down the group, the atomic number and number of shells also increase. 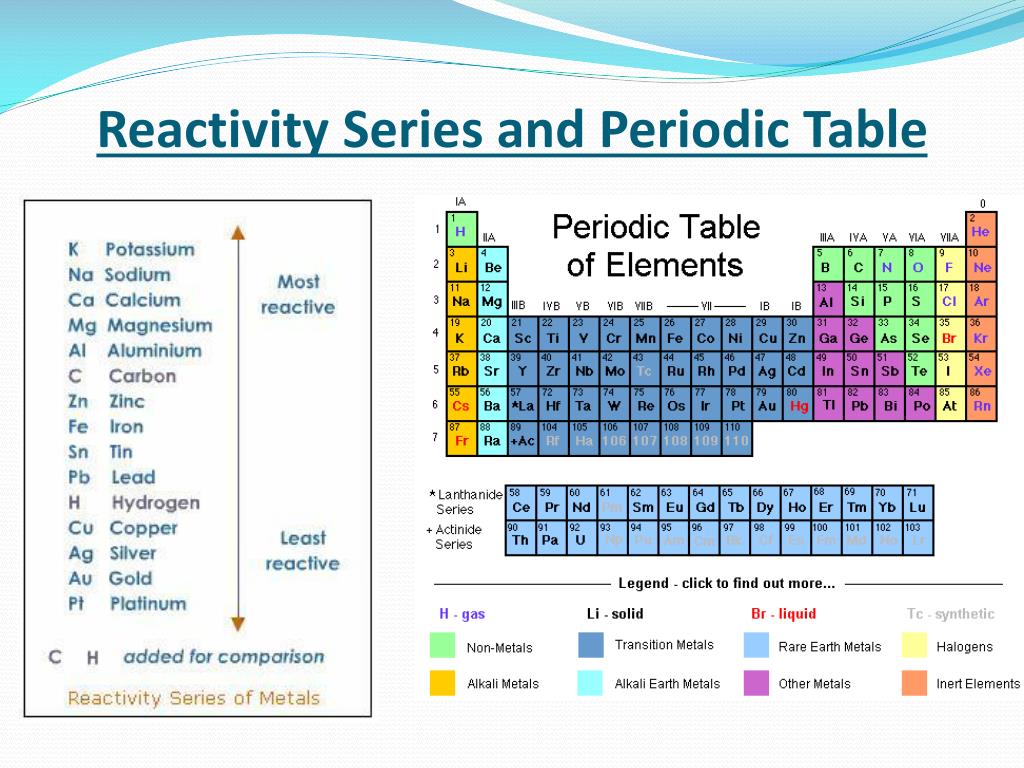
The alkali metals have the largest atomic radii and the lowest first ionization energy in their respective periods.Īlthough all alkali metals have one valence electron in their valence shell, there are slight differences in the reactivity order of elements on going down the group. The properties of the alkali metals are similar to each other as expected for elements in the same family. All alkali metals have their outermost electron in the s-orbital and thus they correspond to the s-block. These elements constitute the first group of the periodic table. The alkali metals are lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr). Alkali metals tend to lose this electron when they participate in reactions. All alkali metals have only one electron in their valence shell. This behavior is in sharp contrast to that of the p-block elements, where the occurrence of two oxidation states separated by two electrons is common, which makes virtually all compounds of the p-block elements diamagnetic.ĭue to a small increase in successive ionization energies, most of the transition metals have multiple oxidation states separated by a single electron.Hint: In chemistry, reactivity is a measure of how readily a substance undergoes a chemical reaction. The occurrence of multiple oxidation states separated by a single electron causes many, if not most, compounds of the transition metals to be paramagnetic, with one to five unpaired electrons. Because of the slow but steady increase in ionization potentials across a row, high oxidation states become progressively less stable for the elements on the right side of the d block. 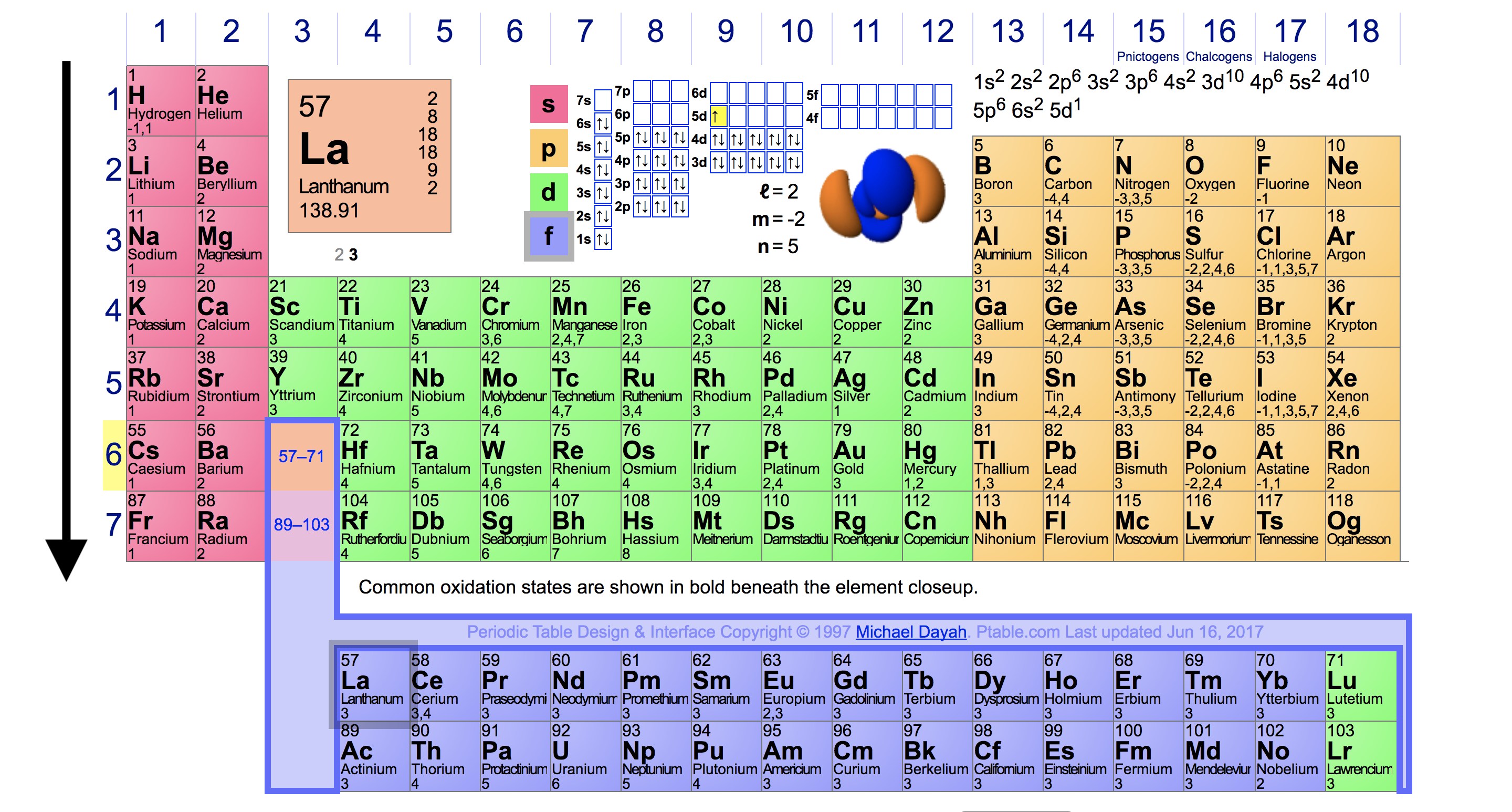
Manganese, for example, forms compounds in every oxidation state between −3 and +7. The relatively small increase in successive ionization energies causes most of the transition metals to exhibit multiple oxidation states separated by a single electron. Thus all the first-row transition metals except Sc form stable compounds that contain the 2+ ion, and, due to the small difference between the second and third ionization energies for these elements, all except Zn also form stable compounds that contain the 3+ ion. This in turn results in extensive horizontal similarities in chemistry, which are most noticeable for the first-row transition metals and for the lanthanides and actinides. The similarity in ionization energies and the relatively small increase in successive ionization energies lead to the formation of metal ions with the same charge for many of the transition metals. /chart-of-periodic-table-trends-608792-v1-6ee35b80170349e8ab67865a2fdfaceb.png)
Trends in Transition Metal Oxidation States As a result, the metals in the lower right corner of the d block are so unreactive that they are often called the “noble metals.” The electronegativity of the elements increases, and the hydration energies of the metal cations decrease in magnitude from left to right and from top to bottom of the d block. 
\): Some Trends in Properties of the Transition Metals. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
